- europages
- >
- Medical tests
- >
- HANGZHOU SINGCLEAN MEDICAL PRODUCTS
- >
- Bacterial Vaginosis Combo Rapid Test Kit CE Approved
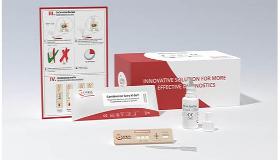
Bacterial Vaginosis Combo Rapid Test Kit CE Approved
Singclean Bacterial Vaginosis Quick Test Kit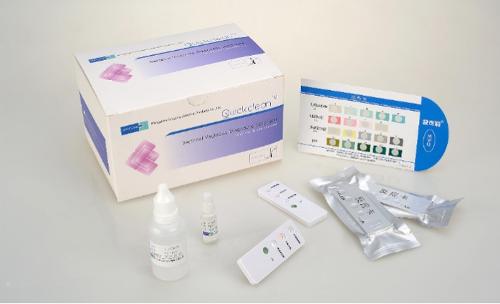
Description
[Product name] Bacterial Vaginosis Quick Test Kit (Sialidase Detection) [Packing size] 20 pcs/kit; 50 pcs/kit [Intended use] For qualitatively testing activity of neuraminidase in female’s vaginal fluid, only for auxiliary diagnosis of bacterial vaginosis. [Testing principle] Neuraminidase is one of pathogenic factors of bacterial vaginosis (BV). Activity of neuraminidase in vaginal fluid of BV women is significantly higher than that in vaginal fluid of normal women; there is no or only a small amount of neuraminidase in vaginal fluid of normal women. By testing for neuraminidase higher than normal level in the vaginal fluid, patients can be diagnosed whether have BV. This kit mainly contains bacterial neuraminidase-specific chromogenic substrate solution.
- Medical tests
- test kit
- Bacterial Vaginosis
- medical test
Product characteristics
- Specification
- 20 pcs/kit; 50 pcs/kit
Similar products

EC PLAZA
South Korea
Our company has been growing for 20 years with the goal of One Health. - Mission: Contribute to human and animal health with diagnostic business. - Vision: Company supplying the world’s highest quality diagnostic kits.
Request for a quote
S.A.M MED BV
Netherlands
Syringe Testing is a critical step in developing and commercializing needlebased injection systems for medical use. By testing mechanical properties, such as breakloose and glide force, as well as biocompatibility, extractables and leachables, sterility, packaging and transit properties, we can determine if the device is within standard specifications, reducing the likelihood of improper function. Eurofins Medical Device Testing network of laboratories has experience performing an array of standard and nonstandard testing on syringes to verify their safety. Choose Eurofins Medical Device Testing to help you Determine the necessary testing requirements for your device Write GMPcompliant and ISO standard driven protocols and methods Perform the required tests to demonstrate that the expected performance criteria are satisfied Complete batch release activities and/or gain regulatory approval
Request for a quote
TRIPLEX INTERNATIONAL BIOSCIENCES
China
TheSARSCoV2 IgG/IgM Rapid Test Kit is a qualitative membrane strip basedimmunoassay for the detection of antibodies (IgG /IgM) to SARSCoV2 inhuman Whole Blood/Serum/Plasma. The test kit consists of1) a burgundy coloredconjugate pad containing SARSCoV2 recombinant envelope antigens conjugatedwith Colloid gold (SARSCoV2 conjugates), 2) a nitrocellulose membrane stripcontaining two test lines (IgG / IgM lines) a nd a control line (C line). TheIgM line is precoated with the Mouse antiHuman IgM antibody, IgG line iscoated with Mouse antiHuman IgG antibody.
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free