- europages
- >
- Clean room systems and processes
- >
- DRESSLER GROUP
- >
- Clean room production (GMP conditions)
Clean room production (GMP conditions)
Clean room production (GMP conditions)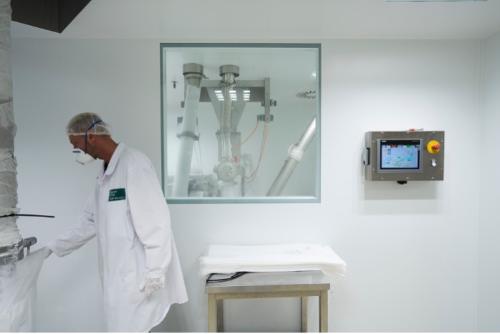
Description
Our production is done in specifically equipped rooms, to rule out the possibility of product contamination from the outside. This includes, for example: special access authorisation, airtight walls and ceilings, windows that cannot be opened, air supply to the rooms being filtered, higher inside air pressure in relation to the outside air, insect lamps to prevent contamination with insects, mandatory protective clothing, shoe covers, gloves and hair covers, written authorisation with documentation for all work or events taking place in the rooms, such as cleaning, conversions, malfunctions. We have specialised in the particular needs of the pharmaceutical, food and health industries. All production takes place under clean room conditions and in compliance with the highest hygiene standards. Some sections are FDA-approved. Food additives are processed in accordance with the HACCP principles and in compliance with the European food hygiene directives and are prepared for dispatch.