- europages
- >
- Medical and surgical instruments
- >
- ASEL MEDIKAL A.S.
- >
- Surgical Instruments
Surgical Instruments
Surgical Instruments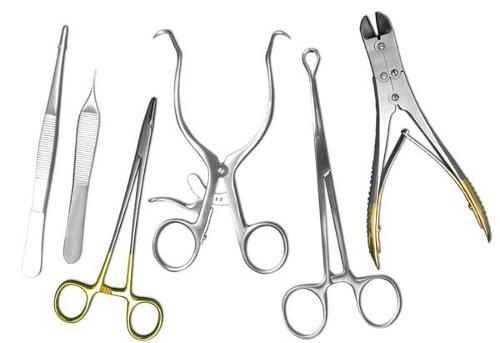
Description
Our products under the title of surgical instruments have the diversity that can meet the needs of surgery departments of health institutions and organizations. Surgical instruments for general surgery, orthopedic surgery, neurosurgery, ENT surgery, cardiovascular surgery, urology, micro-surgery, gynecology, and plastic surgery are produced in our plants.
- Medical and surgical instruments
- surgical instruments
- surgery
- surgical hand tools
Similar products

COMPOSET MEDICAL
Turkey
Non,sterile, autoclavable, sterilizable by gas (Ethylene oxide) kocher forceps. Thanks to its superrior lock it's not easy to open while holding
Request for a quote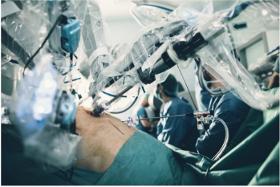
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Robotic surgery is considered a minimally invasive surgery carried out using mechanical arms that have surgical instruments as well as a camera arm attached which are controlled by the surgeon seated at a computer console near the operating table.
Request for a quote
WESTLAKE PLASTICS EUROPE
France
PROPYLUX® HS products deliver the required attributes for orthopedic applications including biocompatibility, chemical resistance and dimensional stability. Launched in 2004 by Westlake Plastics Europe, PROPYLUX® HS has gained wide acceptance by orthopaedic device manufacturers in Europe. At a similar price point as Pomalux, PROPYLUX® HS offers superior dimensional stability performance when processed through repeated 134°C Prion autoclaving cycles.The material also provides excellent chemical resistance to disinfecting and cleaning solutions and exhibits a very good balance of strength, toughness and ductility. As such, PROPYLUX® HS is well suited for use in applications including trial heads and cups for hip implants, knee provisional trials and impactor applications. The PROPYLUX® HS product line consists of 15 colors and 13 diameters. All colored materials are made with FDA compliant ingredients and the shaped rods have been tested and comply with key parts of ISO 10993.
Request for a quote
MPS MICRO PRECISION SYSTEMS AG
Switzerland
Novostia, a Swiss startup founded in 2017, has entrusted MPS with the manufacture of its TRIFLO artificial valve, a revolutionary aortic prosthesis on several levels. Thanks to its aerodynamic design, the presence of three leaflets and the absence of physical pivot axes for the leaflets, the TRIFLO valve aims to satisfy the following three criteria: - A device lifespan at least equal to that of the patient, to avoid the need for a second surgical procedure. This means more than 30 million flap openings and closures per year, with no leaks, blockages, cracks, ruptures or flap escapes. - The artificial valve must not lead to thrombosis, so that patients do not need to take anticoagulants. - Operation (flap opening and closing, blood flow through the valve) must be as silent as possible, so as not to disturb the patient or those around him, day or night. The TRIFLO device is in clinical trial and is not approved for sale.
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free