- europages
- >
- Pharmaceutical products
- >
- VINKEM LABS LIMITED
VINKEM LABS LIMITED
India

Vinkem Labs Ltd. is a leading WHO GMP manufacturer and exporter of niche "difficult to make" Oncology (anti cancer) products from India since 1994 specialised in the field of Natural Product isolation and synthesis of high value, difficult to make cytotoxic Oncology products. Our In-house R&D facility is recognised by department of scientific and industrial research (DSIR) under the ministry of science and technology, Government of India. We also have state of art counter current extractor made of stainless steel having capacity of 60 tonnes /month. We specialise in manufacturing cytotoxic Oncology API Products like Vinorelbine(first and only manufacturer and exporter in India), Vincristine, Vinblastine used in anti-cancer treatment With fresh investments of US$ 15 million, Vinkem Labs is implementing green field, state of the art sterile manufacturing facility designed to USFDA standards for Oncology Injectables.
Website
Unknown
Ah-29, Shanthi Colony, 4th Avenue, Anna Nagar
600040 Chennai - India
Activities of VINKEM LABS LIMITED
- Pharmaceutical products
- Contract Manufacturing
- Cytotoxic Oncology API and Injectable Manufacturer
- Vinblastine
- Vincristine
europages also recommends
A selection of companies related to the activity:
A selection of products that might interest you

EOSWISS PHARMA
Switzerland
We manage investment projects for you, such as production expansions, installation of new plants, greenfield construction projects, brownfield construction projects. We focus on the rapid implementation of your project. With these short project durations, you have low project costs and a high return on investment. We adapt our project management to the very specific circumstances of our clients Contact us for an initial discussion! This makes our project management flexible enough to handle the changes that occur in every project. Our focus is on short "time to market" and thus low project costs. This is the only way for projects to pay off quickly. We execute projects very quickly and achieve significant time savings compared to traditional project management. Pharmaceutical production •After qualification and commissioning, we also take care of the start of production. •Optimisation of the production parameters •Validation •Transfer to routine operation
Request for a quote
EOSWISS PHARMA
Switzerland
We carry out the entire technology transfer or production transfer from a manufacturer to a contract manufacturer (CMO, CDMO) for you. Contact us for an initial discussion! In the process, we look for the right CMO for you and prepare a pre-selection. You decide on the best manufacturer, then we carry out the entire transfer for you: •Planning resources •Timetable including work packages •Structure the cooperation with the CMO •Prepare all necessary documents •technical transfer •Adjusting the production parameters, optimisation •we validate the transferred production process •handed over to routine production We bring all the necessary specialists into the project ourselves, or work hand in hand with your internal team. Depending on how it is needed. We carry out transfers very quickly. This shortens the project duration and reduces your costs: They are quickly back on the market! This reduces transfer costs in the long term. Contact us for an initial discussion!
Request for a quote
COMFORTPAT B.V.
Netherlands
Indication : Herbal medicine for catarrhal diseases of the respiratory tract. The drug is used to improve the symptoms of chronic inflammatory bronchial diseases; Acute inflammation of the airways accompanied by coughing. A notice: If the symptoms persist or if you experience shortness of breath, fever or purulent or bloody sputum, you should see a doctor immediately.
Request for a quote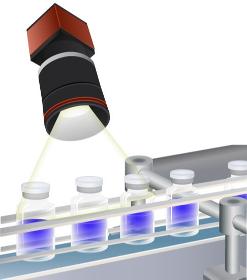
EOSWISS PHARMA
Switzerland
We support your manufacturing with our polarised light camera and analytical software. We film production issues of transparent material (vials, blisters, glass items, foils) with polarized light. Defects, scratches, flaws, residual tensions brought in during manufacturing become visible. Hence, they can be corrected and improved. You receive an improved manufacturing process, with higher quality and higher capacity.
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free



