- europages
- >
- Pharmaceutical products
- >
- AGON PHARMA GMBH
AGON PHARMA GMBH
Germany
Distributor

Product catalogue preview
AGON is a service provider with its own laboratories, environmental chambers and climate cabinets. AGON is based in Wendlingen – only 15 minutes from Stuttgart airport. AGON has more than 20 years of experience in the development and approval of plant-based and chemical-based medical drugs on the basis of over 300 development processes, approval processes and post-approval processes. Furthermore, AGON develops medical products to order as well as supplementary balanced diets and nutritional supplements.
Website
Documents
Company info
Key figures
-
- Company headcount
- 11 – 50
Organisation
-
- Year established
- 1988
-
- pages.epage-home.site-status
- Registered office – Parent company
-
- Main activity
- Distributor
Business info
Trading areas
- International
Activities of AGON PHARMA GMBH
- Pharmaceutical products
- Pharmaceuticals - basic and auxiliary products
europages also recommends
A selection of companies related to the activity:
A selection of products that might interest you

EOSWISS PHARMA
Switzerland
We carry out the entire technology transfer or production transfer from a manufacturer to a contract manufacturer (CMO, CDMO) for you. Contact us for an initial discussion! In the process, we look for the right CMO for you and prepare a pre-selection. You decide on the best manufacturer, then we carry out the entire transfer for you: •Planning resources •Timetable including work packages •Structure the cooperation with the CMO •Prepare all necessary documents •technical transfer •Adjusting the production parameters, optimisation •we validate the transferred production process •handed over to routine production We bring all the necessary specialists into the project ourselves, or work hand in hand with your internal team. Depending on how it is needed. We carry out transfers very quickly. This shortens the project duration and reduces your costs: They are quickly back on the market! This reduces transfer costs in the long term. Contact us for an initial discussion!
Request for a quote
COMFORTPAT B.V.
Netherlands
Indication : Herbal medicine for catarrhal diseases of the respiratory tract. The drug is used to improve the symptoms of chronic inflammatory bronchial diseases; Acute inflammation of the airways accompanied by coughing. A notice: If the symptoms persist or if you experience shortness of breath, fever or purulent or bloody sputum, you should see a doctor immediately.
Request for a quote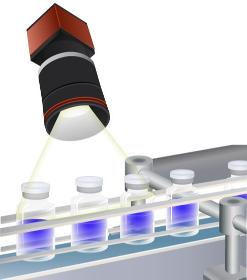
EOSWISS PHARMA
Switzerland
We support your manufacturing with our polarised light camera and analytical software. We film production issues of transparent material (vials, blisters, glass items, foils) with polarized light. Defects, scratches, flaws, residual tensions brought in during manufacturing become visible. Hence, they can be corrected and improved. You receive an improved manufacturing process, with higher quality and higher capacity.
Request for a quote
EOSWISS PHARMA
Switzerland
We plan and perform your complete process validation. From the first gap analysis and risk management, planning, execution and reporting. We support you with full project management, providing support where needed. You free your ressources for your projects, we take care for validations and all connected activities. You reach your goals safely, quicker and faster.
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free



