- europages
- >
- Medical Equipment
- >
- SHENZHEN CREATIVE INDUSTRY CO., LTD.
SHENZHEN CREATIVE INDUSTRY CO., LTD.
China
Manufacturer/ Producer


SHENZHEN CREATIVE INDUSTRY CO., LTD.
China
NEO ECG is the latest release model of ECG, tablet design, compact in size with low weight, color touch screen, easy to operate and carry. Support synchronous acquisition and display of 9/12/15/18 leads ECG waveform. Support connect to AI ECG Platform.
Request for a quote
SHENZHEN CREATIVE INDUSTRY CO., LTD.
China
AI View Patient Monitor, tilted, colorful & touch screen with high resolution. Complete parameter, support 3/5/6/10 eclectrode ECG, SpO2, Pulse Rate, BP, TEM, EtCO2, Dual IBP monitoring. Provide EWS function, to predict the risk level of the patient's physiological state by combining parameters. Support connect to AI ECG Platform. AI ECG Platform: Test by 1,000,000 independent MIT test data, the average accuracy rate of the AI-ECG platform reach 95.2%. High Analysis Speed, take 1s for automatically resting ECG analysis. Support 16 types of cardiac classification, 104 types of ECG diagnostic classifications.
Request for a quote
SHENZHEN CREATIVE INDUSTRY CO., LTD.
China
C3/C3PluS Diagnostic Ultrasound System As a new member of Sonolepu portable color Doppler ultrasound family, C3/C3Plus is a compact and ultra-light ultrasound system which has taken into consideration of all the necessary clinic requirements and needs in order to enable ultrasound users to provide better patient care.
Request for a quote
SHENZHEN CREATIVE INDUSTRY CO., LTD.
China
ECG-1112M is an Electrocardiograph which can collect 12-lead ECG signal and print waveform by the thermal printing system. 1.180 ECG files can be saved in the ECG 2.Over 5000 ECG files can be saved to SD card(optional) 3.Freeze, Pre-10-second print and Trigger print function to observe any abnormal ECG waveforms 4.Quick acquisition and stability, waveforms get stable within 3s 5.HR detection alarm and pacemaker detection supported 6.Cine-loop function: saved ECG waveform can be played back 7.Auto-save function: users can choose whether to automatically save the files with the printout or not 8.Support magnetic card reader, bar code scanner, transferring information conveniently 9.Support external laser printer to achieve A4 size report output 10.Data management software on PC(optional) 11.Reliable automatic measurement & Interpretation tested with CSE&AHA database
Request for a quoteShenzhen Creative Industry Co., Ltd. is a professional medical devices manufacturer and marketer founded in January 2000. With 21 years of experience in the industry, we have been providing Patient Monitors, Pulse Oximeters, Homecare Devices and Rehabilitation Systems to thousands of users all over the world. In 2009, we joined the Lepu Medical which is the top 2 medical giant in the medical industry listed company. We’re very pleased to take this opportunity to recommend our products to you . Attached is the brochure of our newest AI monitor, pls kindly check.We are able to provide the best prices, the highest quality and the superior service. Looking forward to your reply for further discusstion.
Website
Documents
Manufacturer/ Producer
Xili Street
SONGBAI ROAD Shenzhen - China
Company info
Key figures
-
- Company headcount
- > 500
-
- Sales staff
- 11 – 50
-
- % of export sales
- 70%
Organisation
-
- Year established
- 2000
-
- pages.epage-home.site-status
- Registered office – Parent company
-
- Main activity
- Manufacturer/ Producer
Business info
Trading areas
- International
Delivery areas
Activities of SHENZHEN CREATIVE INDUSTRY CO., LTD.
- Medical Equipment
- hypertension
europages also recommends
A selection of companies related to the activity:
A selection of products that might interest you

ANSMANN AG
Germany
The new IPC charger series is the latest innovation in the field of quick-charging. The universal chargers have integrated communication and support all popular communication protocols, such as CAN, I2C, SM, LIN, HDQ. Li-ion rechargeable battery packs from 24 V to 48 V will be automatically detected. Once the E-BUS charger has made contact with the rechargeable battery, an extensive diagnostics procedure is started. The intelligent chargers identify the rechargeable battery voltage, the charge current is adapted and the charge process started. The output power here is up to 300 W. This enables the shortest possible charge times with simultaneous exchange of parameters to be realised with applications such as in drive systems, power tools or medical equipment. With the IPC 300W we are delighted to present a universal charger that can adapt to all lithium ion rechargeable battery systems.
Request for a quote
ANSMANN AG
Germany
Planning a new development or a redesign of a rechargeable battery or charger? ANSMANN – Your partner from the initial idea through the planning, design, development and manufacturing stages and on to serial manufacturing. Based on your application and the requirements, we can guide you with professional advice for the selection of the correct cells, the capabilities of the necessary protective circuitry, compliance with standards and certification (as early as in the development phase), the first prototypes as STL parts and the first measurements. We can support you through all of the important steps towards your final decision. We can take on the design and manufacturing of the housing parts and the injection and stamping tools required for this. Single-source delivery and manufacturing guarantees optimum interaction between rechargeable battery, BMS and charger. The cells that we use are delivered by renowned manufacturers such as Panasonic, LG, Samsung, Sanyo, A123, Sony, etc.
Request for a quote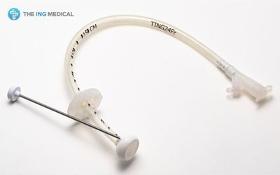
EC PLAZA
South Korea
1) Product Description The Gastrostomy Feeding Tube are indicated for use in patients who require long term feeding, are unable to tolerate oral feeding, who are at low risk for aspration, require gastric decomparession. It is a tube used to supply food and drugs to the stomach. 2) Product Features - Easy and less traumatic to insert due to the soft and compressible internal dome. - Radiopaque dome allows x-ray placement confirmation - Identification of tube obstructions enhanced with translucent tubings - Soft, less traumatic silicone designed for patient comfort
Request for a quote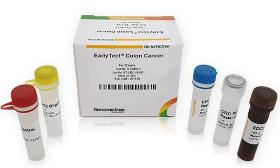
EC PLAZA
South Korea
Product Description EarlyTect Colon Cancer Test was approved by the Korea Ministry of Food and Drug Safety through clinical trials at Yonsei Medical Center, Severance Hospital, and Severance Check-up Center for eligible subjects, 585 men and women aged 30 to 80. Clinical trials using fecal DNA demonstrated that CRC can be diagnosed with 90% sensitivity and specificity. EarlyTect Colon Cancer Test is a qualitative real-time PCR test that measures methylated Syndecan-2, a biomarker clinically related to precancerous lesions of CRC, in stool DNA to diagnose colorectal cancer. This product is not intended to confirm colorectal cancer, so if a subject was positive, CRC or precancerous lesions of CRC may exist. Product Standard - Cancer Type: Colorectal Cancer - Subject: Subjects for asymptomatic colorectal cancer screening - Biomarker: SDC2 me The price and transaction conditions are negotiable. Please Request a Quote for the transaction
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free



