- europages
- >
- Medical tests
- >
- HANGZHOU SINGCLEAN MEDICAL PRODUCTS
- >
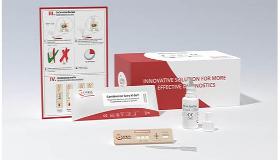
- Monkeypox virus IgG/IgM Antibody Test kit with CE
Monkeypox virus IgG/IgM Antibody Test kit with CE
Singclean Monkeypox virus Antibody Test kit(Immunochromatography)
Description
Monkeypox virus IgG/IgM Test Kit (Immunochromatography) is a solid phase immunochromatographic assay for the rapid, qualitative detection of IgG/IgM antibody to Monkeypox virus in human blood. This test provides only a preliminary test result. Therefore, any reactive specimen with the Monkeypox virus IgG/IgM Test Kit (Immunochromatography) must be confirmed with alternative testing method(s) and clinical findings.
- Medical tests
- antibody test kit
- monkeypox
- diagnosis
Product characteristics
- Specification
- 1 test/box, 5 tests/box, 20 tests/box
- SAMPLE
- Human serum, plasma or whole blood
Similar products

TRIPLEX INTERNATIONAL BIOSCIENCES
China
The kit is used for in vitro qualitative detection of monkeypox virus DNA in blood, wound or pus samples from suspected monkeypox patients. It is for professional use only. Not for self-testing.
Request for a quote
EC PLAZA
South Korea
Our company has been growing for 20 years with the goal of One Health. - Mission: Contribute to human and animal health with diagnostic business. - Vision: Company supplying the world’s highest quality diagnostic kits.
Request for a quote
S.A.M MED BV
Netherlands
Syringe Testing is a critical step in developing and commercializing needlebased injection systems for medical use. By testing mechanical properties, such as breakloose and glide force, as well as biocompatibility, extractables and leachables, sterility, packaging and transit properties, we can determine if the device is within standard specifications, reducing the likelihood of improper function. Eurofins Medical Device Testing network of laboratories has experience performing an array of standard and nonstandard testing on syringes to verify their safety. Choose Eurofins Medical Device Testing to help you Determine the necessary testing requirements for your device Write GMPcompliant and ISO standard driven protocols and methods Perform the required tests to demonstrate that the expected performance criteria are satisfied Complete batch release activities and/or gain regulatory approval
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free