- europages
- >
- Medical Equipment
- >
- KERNEL MEDICAL EQUIPMENT CO., LTD
KERNEL MEDICAL EQUIPMENT CO., LTD
United States
Manufacturer/ Producer

Product catalogue preview

KERNEL MEDICAL EQUIPMENT CO., LTD
United States
Maximum Efficiency The special ultraviolet lamp produced by the Philips company is used as the treatment light source, which has the characteristics of high stability and long life; Dust and Waterproof Dustproof and waterproof grade reachesIP22, so that patients can use it at home more at ease AC power supply The working votage ranges from AC100V to AC240V, 50Hz/60Hz. LCD screen adjustment The viewing direction of the LCD screen can be adjusted according to the needs of use. Timer function Microcomputer timing function, reminder of remaining treatment time, eliminating the trouble of time calculation, and convenient for users to grasp the treatment situation in time. Unique comb design It 's not only suitable for the irradiation of the face, limbs and other parts, the unique comb design can also take into account the treatment of the scalp.
Request for a quote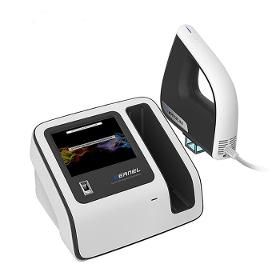
KERNEL MEDICAL EQUIPMENT CO., LTD
United States
Main Features Small size, low cost of consumables,. Quick start, one key output, easy to operate; 8"touch screen combined with humanized GUI design, built-in minimum erythema dose(MED) test function. Treatment handle works with multi-specification-available shading sheet,realized a precise control of the irradiation range, suitable for different sizes of lesion treatment. Good targeting, no harmless to normal skin, easy to treat the lesions where the traditional light hard to reach Dose and time two kinds of work mode,one machine for multiple use. Fingerprint management system and irradiation intensity calibration system, ensure the accurate treatment dosage.
Request for a quote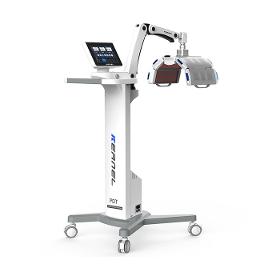
KERNEL MEDICAL EQUIPMENT CO., LTD
United States
PDT machine suitable for acne treatment,elimination of inflammation,and promotion of skin wound healing KN-7000D LED light therapy adopts high-power SMD matrix light source, with high arrangement density, higher irradiation intensity and more uniform light spot; Based on aerodynamic system design, supplemented by super-strong thermal materials to ensure LED light therapy treatment safe, reliable, and stable use in harsh environment; Single color LED light, two colors LED light, and three colors light all can be combined in one treatment head, and multiple LED wavelengths also can be selected, to meet different demands of clinical use; Free-designed cantilever makes the light panel can be fixed and stayed in any angle and position. Kernel PDT device adopts 8" full touch screen and user-friendly GUI design to make the operation more concise and smooth; Unique dose mode and radiation intensity calibration to ensure the precision of the treatment; KN-7000
Request for a quote
KERNEL MEDICAL EQUIPMENT CO., LTD
United States
High power SMD LED light source, 3D simulation matrix arrangement, high intensity, more uniform light spot Various combination of light source, meeting diversified clinical needs Multi light source custom cycle irradiation mode, one-key to complete the system treatment Free telescopic cantilever system, the light source can stay at any position and angle AI intelligent temperature control system ensures safety of treatment Pre-stored 5 treatment plans, one-key selection, making the orperation more convenient Real time intensity detection function Continuous irradiation mode and pulse irradiation mode available, meeting different clinical needs Double safety protection of button switch and power-on password
Request for a quoteThe company KERNEL MEDICAL EQUIPMENT CO., LTD, is a Manufacturer/ Producer, founded in 1997, which operates in the Medical Equipment industry. It also operates in the uv phototherapy, psoriasis phototherapy, led light therapy, and colposcopes industries. It is based in New york,, United States.
Other companies in the same industry:
Website
Documents
Manufacturer/ Producer
228 PARK AVE S #79525
10003 New york, - United States
Company info
Key figures
-
- Company headcount
- 201 – 500
-
- % of export sales
- 55%
Organisation
-
- Year established
- 1997
-
- pages.epage-home.site-status
- Registered office – Parent company
-
- Main activity
- Manufacturer/ Producer
Activities of KERNEL MEDICAL EQUIPMENT CO., LTD
- Medical Equipment
- uv phototherapy
- psoriasis phototherapy
- led light therapy
- colposcopes
europages also recommends
A selection of companies related to the activity:
A selection of products that might interest you

ANSMANN AG
Germany
The new IPC charger series is the latest innovation in the field of quick-charging. The universal chargers have integrated communication and support all popular communication protocols, such as CAN, I2C, SM, LIN, HDQ. Li-ion rechargeable battery packs from 24 V to 48 V will be automatically detected. Once the E-BUS charger has made contact with the rechargeable battery, an extensive diagnostics procedure is started. The intelligent chargers identify the rechargeable battery voltage, the charge current is adapted and the charge process started. The output power here is up to 300 W. This enables the shortest possible charge times with simultaneous exchange of parameters to be realised with applications such as in drive systems, power tools or medical equipment. With the IPC 300W we are delighted to present a universal charger that can adapt to all lithium ion rechargeable battery systems.
Request for a quote
ANSMANN AG
Germany
Planning a new development or a redesign of a rechargeable battery or charger? ANSMANN – Your partner from the initial idea through the planning, design, development and manufacturing stages and on to serial manufacturing. Based on your application and the requirements, we can guide you with professional advice for the selection of the correct cells, the capabilities of the necessary protective circuitry, compliance with standards and certification (as early as in the development phase), the first prototypes as STL parts and the first measurements. We can support you through all of the important steps towards your final decision. We can take on the design and manufacturing of the housing parts and the injection and stamping tools required for this. Single-source delivery and manufacturing guarantees optimum interaction between rechargeable battery, BMS and charger. The cells that we use are delivered by renowned manufacturers such as Panasonic, LG, Samsung, Sanyo, A123, Sony, etc.
Request for a quote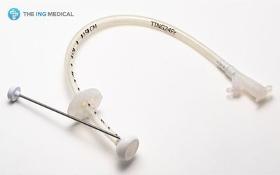
EC PLAZA
South Korea
1) Product Description The Gastrostomy Feeding Tube are indicated for use in patients who require long term feeding, are unable to tolerate oral feeding, who are at low risk for aspration, require gastric decomparession. It is a tube used to supply food and drugs to the stomach. 2) Product Features - Easy and less traumatic to insert due to the soft and compressible internal dome. - Radiopaque dome allows x-ray placement confirmation - Identification of tube obstructions enhanced with translucent tubings - Soft, less traumatic silicone designed for patient comfort
Request for a quote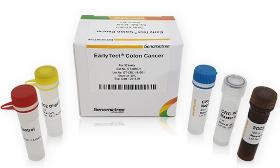
EC PLAZA
South Korea
Product Description EarlyTect Colon Cancer Test was approved by the Korea Ministry of Food and Drug Safety through clinical trials at Yonsei Medical Center, Severance Hospital, and Severance Check-up Center for eligible subjects, 585 men and women aged 30 to 80. Clinical trials using fecal DNA demonstrated that CRC can be diagnosed with 90% sensitivity and specificity. EarlyTect Colon Cancer Test is a qualitative real-time PCR test that measures methylated Syndecan-2, a biomarker clinically related to precancerous lesions of CRC, in stool DNA to diagnose colorectal cancer. This product is not intended to confirm colorectal cancer, so if a subject was positive, CRC or precancerous lesions of CRC may exist. Product Standard - Cancer Type: Colorectal Cancer - Subject: Subjects for asymptomatic colorectal cancer screening - Biomarker: SDC2 me The price and transaction conditions are negotiable. Please Request a Quote for the transaction
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free



