- europages
- >
- Medical tests
- >
- ASCEND CARE LTD
- >
- Sugentech SGTI Flex COVID Antigen Lateral Flow Test x2
Sugentech SGTI Flex COVID Antigen Lateral Flow Test x2
Self Test Kits x 2
Description
Sugentech SGTi-flex COVID-19 Ag is an immunoassay for qualitative detection of Nucleocapsid protein antigen from SARS-CoV-2 in nasopharyngeal and nasal swab specimen. • Specimen: Nasopharyngeal (or Nasal) swab • One Step, Fast results within 15 min • Full package for the whole test procedure, No need for additional tool/device - Swab - Buffer - Test Cassette • Package: 2 test kits *all prices are ex vat * shipping is free on orders over 1000 units to one location Product No. CAGT001E0 Description COVID-19 Antigen Lateral Flow Test. For self-testing only. Approvals CE Marked, MHRA registered Intended Use An immunoassay for qualitative detection of Nucleocapsid protein antigen from SARS-CoV-2 in nasal swab specimen. Accuracy Overall Accuracy Sensitivity Specificity 97.35% 95.07% 99.38% Package Each test is individually boxed Specimen Nasal Swab Storage 2-30 Celsius Test Time 15-30 minutes CE Marked US FDA MHRA Compliant European Commission
- Medical tests
- covid19
- lateral flow tests
- ivd diagnostics
Similar products

EC PLAZA
South Korea
MEDI_SPRAY is classified as a food product and is harmless to the human body, but it is the only technology that inactivates and kills viruses, including corona. [Product Features] Easy portability Ingredients safe for all ages FDA OTC Registration Covid-19 over 99.9% inactivation tested 5 Bacteria over 99.9% inactivation tested [Company Information] MEDIPLUS Mediplus is a prebiotics company that has secured the world's best source technology for yeast and lactobacillus. It is a company that specializes in technology management that develops and distributes various products by utilizing Prebiotics. We are commercializing yeast and lactobacillus-related research products as products that can directly help consumers' real lives, and expanding the scope of business and product coverage by developing various products that help human health. Website : http://www.mediwater.co.kr
Request for a quote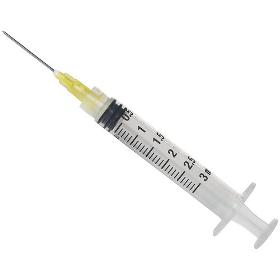
S.A.M MED BV
Netherlands
Syringe Testing is a critical step in developing and commercializing needlebased injection systems for medical use. By testing mechanical properties, such as breakloose and glide force, as well as biocompatibility, extractables and leachables, sterility, packaging and transit properties, we can determine if the device is within standard specifications, reducing the likelihood of improper function. Eurofins Medical Device Testing network of laboratories has experience performing an array of standard and nonstandard testing on syringes to verify their safety. Choose Eurofins Medical Device Testing to help you Determine the necessary testing requirements for your device Write GMPcompliant and ISO standard driven protocols and methods Perform the required tests to demonstrate that the expected performance criteria are satisfied Complete batch release activities and/or gain regulatory approval
Request for a quote
DISERA MEDICAL EQUIPEMENT
Turkey
1ml - 2ml - 3ml - 4ml - 6ml - 9ml - 10ml Vacusera Serum Clot Activator Tubes are used for clinical biochemistry and immunology. The inner wall of the Serum Clot Activator Tube is specially coated with clot activator, which activate coagulation process. Vacusera Serum Separator Tubes contain inert gel; during centrifugation, gel forms a stable barrier between serum and blood cells.
Request for a quote
AXON CABLE SAS
France
Axon' has developed picocoax® a complete range of miniature coaxial cables and harnesses for applications where a large number of signals have to be transmitted with no interference. They can be used for measurement equipment and medical devices. Picocoax miniature coaxial cables offer a good compromise between small diameter (for example 0.20 mm) and capacitance of 50 to 100 pF/m. Picocoax® miniature coaxial cables are made with silver plated copper alloy conductor and high performance dielectric materials including FEP or Celloflon (porous PTFE patented by Axon'). Axon' also offers multipicocoax made with bundles of 18 to 304 picocoax cables. These composite cables are very flexible.
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free