- europages
- >
- Medical Equipment
- >
- ALFA-MED LLC
- >
- products
ALFA-MED LLC - Medical equipment for health assessment manufacturer
Russia
Multi-Category


ALFA-MED LLC
Russia
SensitivE Audit® devices are the premium segment in the range of SensitivE diagnostic tools. The device uses the advanced LifeStream® program and is able to run complete diagnostics of a human body in a short space of time, without any harm to health. SensitivE Audit has the form of a transformer that allows you to supplement and improve the basic set with different modules, as well as a greater number of reference processes with corrected frequency spectra, which provides for substantially increasing testing accuracy. The accuracy of the device SensitivE Audit diagnostics reaches — 97% SensitivE Audit was inspired by cutting-edge high-speed technologies and has been globally recognized by the scientific community.
Request for a quote
ALFA-MED LLC
Russia
The work of the TempoHeart module is based on the method of scoring the heart rate vegetative regulation. This method is popular due to its high information value and non-invasiveness. Cannot be used without an interface block. The module can: • evaluate the heart rate autonomic regulation; • to predict in advance (to prematurely reveal) the hazards of a myocardial infarction, hypertensive crises and strokes; • to determine the metabolic and electrolyte disorders in the myocardium; • identify risk groups for heart rate increased stability; • to make a cold evaluation of the autonomic nervous system's reaction when exposed to electromagnetic fields, intoxications and other pathogenic factors; • to assess the activity of the respiratory component when measuring heart rate variability, recording the parameters of blood movement in the vessels; • to expose the stress resistance level.
Request for a quote
ALFA-MED LLC
Russia
Carries out spectrophotometric analysis with the registration of arterial temperatures infrared radiation. Cannot be used without an interface block. The module can: • noninvasively obtain 85 biochemical parameters of peripheral and hemodynamic blood composition; • obtain hemodynamic parameters; • obtain various information about the oxygen transportation in the blood; • analyze the cardiovascular system activity; • study affiliation with the hematopoietic, immune and enzyme systems of the body.
Request for a quote
ALFA-MED LLC
Russia
SensitivE Audit® DynamicB The module allows analyzing tissue conductivity and microcirculation disorders using certain dosed stimuli. Cannot be used without an interface block. The module can: • automatically conduct an incremental scanning of the head and limbs with impulses of negative and positive polarity (20 derivations), with a repetition rate of 10 Hz. At that, the electric signal test is physiological and does not cause discomfort; • to readjust the signal received via the use of direct or alternating current influence in the measuring range to exclude effect of fabric electrolysis; • use sequential transfer of frequency-wave information to the object under study along the diagonals, in accordance with the distribution of energy meridians; • to carry out early prediction of neoplastic and fibrodystrophic processes in selective foci; • to reveal the hidden foci of tissue conduction disorders and their influence on the studied object's functional reserves of adaptation.
Request for a quote
ALFA-MED LLC
Russia
Body composition measurement by means of the LineBIA method is based on assessing the distribution of water volumes in the human body and on tissue ability to conduct electricity. Cannot be used without an interface block. The module can: • determine the quantitative content of fluid in the body, body mass index, standard metabolism; • determine the active cell mass, bone mass, fat mass; • obtain unbiased data on the human body quantitative composition; • get linear control and timely correction of nutrition programs in clinical practice and sports medicine; • an unbiased assessment of physical conditioning and nutrition; • analyze the water-salt metabolism; • analyze the state of water-salt metabolism during the period of adaptive changes in the human body, under changing weather conditions (mountain climate, hot climate, or dry climate).
Request for a quote
ALFA-MED LLC
Russia
NeuroStim is a method that increases the level of health and treatment quality by means of chromotherapy and auditive stimulation. Cannot be used without an interface block. The module can: • boost and improve memory; • raise the intelligence level; •improve physical endurance; •improve mental activity; • increase the creative potential; •activate study skills; •even out the psycho-emotional state; •improve and regulate sleep; •carry out recovery after loads (emotional, physical); • undertake measures against stress; • to improve the psychophysiological condition of an athlete, to rationally prepare for competitions; •assist in conducting breathing sessions, meditations; •help to relax and energize.
Request for a quote
ALFA-MED LLC
Russia
Sensitiv imago® 530 is the previous model in the SensitivE line. Hi-tech hardware-software complexes for diagnostic scan, analysis and healing of organism. Senstiv imago 530 device is being assembled according to the new European quality management standards 9001 full accepted in EU in 2013. The device passed the medical laboratory testing class 2, according to the European Directive.
Request for a quoteeuropages also recommends
A selection of companies related to the activity:
A selection of products that might interest you

SAM3D
France
In addition to the Sam3D software, the following equipment will be used to carry out the postural assessment assisted by 3D imaging Sam3D TOWER image acquisition device 24" All In One computer Sam3D TOWER software Sam3D reference ground plate Marking pen Sam3D Roll-Up or Sam3D Wall Adhesive
Request for a quote
SAM3D
France
Marking key points on the body - Photographs of different positions - A personalised assessment is given to each patient
Request for a quote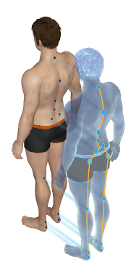
SAM3D
France
SAM3D LITE/LITE PLUS is a powerful, easy-to-use and intuitive posture analysis software package. Thanks to its modern, user-friendly interface, carrying out a postural assessment has never been easier! Its clear structure will guide you through the analysis process right up to the printout. 3D reconstruction of the whole body in a functional standing position. Non-contact 3D measurement in all three planes: - Frontal - Sagittal - Transverse Whole body analysis 3D reconstruction of the spine and the whole body The points placed on the skin by the practitioner enable the Sam3D system to reconstruct the patient's anatomical the patient's anatomical segments with great precision.
Request for a quote
SAM3D
France
To accompany the Sam3D Lite or Lite+ software, a kit is available for carrying out postural diagnostics in a mobile version. The kit includes: Sam3D LITE image acquisition device Carrying case Sam3D reference ground plate Compact tripod Sam3D LITE / LITE+ analysis software Connection cable Tape measure Marking pen
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free



